When Time Runs Out for the Heart:
Low NAD+ Levels Disrupt the Body’s Internal Clock During Cellular Aging
Declining NAD+ levels in the aging heart weaken circadian rhythm and cardiac health
Countless processes in the body – including cellular metabolism and the regulation of gene expression – follow an internal clock that responds to and anticipates environmental and behavioral cues. These phenomena, known as circadian rhythms, become weaker in the aging organism. The resulting desynchronization between tissues within the body, as well as between the organism and its environment, can accelerate the development of diseases such as chronic heart failure. Scientists at the Max Planck Institute for Heart and Lung Research have now demonstrated that NAD+ levels in the heart decline with age and that NAD+ levels are directly linked to the health of the internal clock. Increasing or maintaining NAD+ levels through dietary supplements could therefore represent a therapeutic strategy for preventing age-related diseases.

Circadian rhythms are patterns of behavior – such as feeding/fasting or sleep/wake cycles – that occur in coordination with daily external cues like the light/dark cycle. The molecular basis of these rhythms is maintained by the cells themselves, which then cooperate within an organ to regulate tissue-specific functions. This coordination extends throughout the entire organism, synchronizing countless functions across the body and with the environment. With increasing age, both the molecular foundations and physiological processes begin to decline and shift, leading to desynchronization and, consequently, an increased likelihood of various chronic diseases such as heart failure.
Scientists from the research group “Circadian Rhythms of Cardiac Metabolism” at the Max Planck Institute for Heart and Lung Research in Bad Nauheim have now clarified important links between heart health and the regulation of rhythmicity during aging, and have published their study in the journal Communications Biology.
“Every heartbeat is more than just a mechanical impulse – it is a finely tuned physiological process controlled by the internal clock,” says Pieterjan Dierickx, head of the research group.
In the cardiovascular system, the internal clock regulates, among other things, energy metabolism, heart rate, and ion signaling.
“Our research therefore focused on NAD+. This is a molecule that plays a central role in cellular energy metabolism,” explains Bryce Carpenter, first author of the study.
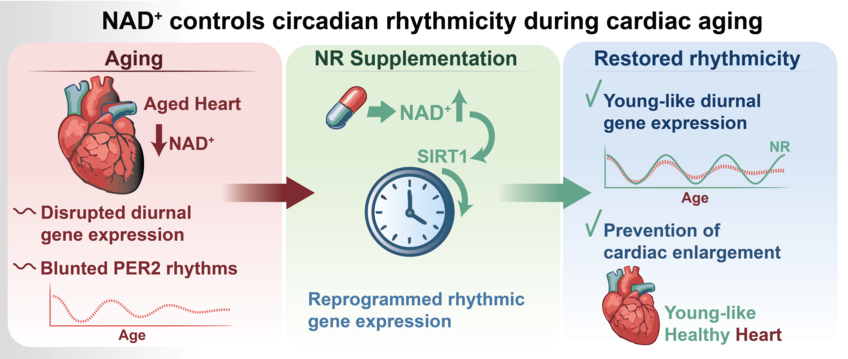
The two researchers had previously found that NAD+ is also rhythmically available in the heart, suggesting that the molecule acts as a key link between rhythmicity and metabolism. They have now discovered, however, that NAD+ levels in the heart decrease with age, in parallel with the weakening of the internal clock.
To further investigate the cause of the decline in oscillating gene expression, the scientists treated aged mice with an NAD+ precursor molecule. “This pharmacological increase in NAD+ levels restored oscillating gene expression in old mice and more closely resembled the patterns observed in young mice,” explains Dierickx.
The researchers in Bad Nauheim then examined the effects on the animals’ heart health: older mice with elevated NAD+ levels not only showed improved rhythmic gene expression, but also a reversal of pathological cardiac changes—such as an enlarged heart muscle. “By increasing NAD+ levels, the old mice returned to a more youthful state,” explains Dierickx. Further experiments will investigate how NAD+ could serve as a therapeutic target, as the researchers are exploring potential mechanisms that benefit from increased NAD+ levels, such as the protein SIRT1. “Ideally, we will find a way to stabilize the internal clock during aging and thereby halt the development of age-related diseases,” says Dierickx.