Joint research work on pulmonary hypertension awarded
Christoph Zehendner and Chanil Valasarajan awarded by René Baumgart Foundation
The joint work “Long Noncoding RNA TYKRIL Plays a Role in Pulmonary Hypertension via the p53 mediated Regulation of PDGFRβ” of Christoph Zehendner and Chanil Valasarajan is the research award winner 2021 of the René Baumgart Foundation. Chanil Valasarajan is a PhD candidate at our institute and conducts research at the lab of Soni Pullamsetti. Chair of the Foundation and Direcor of the Department 'Lung Development and Remodelling' is Prof. Werner Seeger.
This work identifies the previously uncharacterized long noncoding RNA TYKRIL as an important signaling molecule for the pathogenesis of pulmonary arterial hypertension (PAH) through regulation of the p53/PDGFRβ signaling axis. TYKRIL may represent a relevant target molecule for future novel PAH therapeutic strategies.
Pulmonary arterial hypertension (PAH, pulmonary hypertension group 1) is a disease of the pulmonary circulation in which remodeled pulmonary arteries increase pulmonary vascular resistance and can ultimately lead to right heart failure, which is very distressing for patients. Remodeling of the pulmonary artery in PAH is mainly characterized by increased growth of various cells such as pulmonary artery smooth muscle cells (PASMCs), with other cells such as pericytes contributing as well. Various treatments for PAH exist, but so far no optimal results have been achieved. In particular, in the field of PAH, there is so far limited knowledge about new signaling molecules and their molecular mode of action, which play a role in all cell types relevant to PAH pathogenesis (PASMCs, fibroblasts, endothelial cells, and pericytes). Therefore, there is a need to identify molecules that have relevance in all these cell types that drive vascular remodeling and to explore their therapeutic potential as well as their molecular mode of action.
Long non-coding RNAs (lncRNAs) are relevant in the regulation of a variety of biological processes. However, their role in the pathogenesis of PAH is still unclear. To investigate the role of lncRNAs in PAH, a cross-institutional collaboration was established, in particular between the institutes of the two first authors of the paper, the Institute of Cardiovascular Regeneration (Christoph Zehendner, from the research group of Prof. Stefanie Dimmeler), Goethe University Frankfurt and the Max Planck Institute for Heart and Lung Research Bad Nauheim (Chanil Valasarajan from the research group of Prof. Soni Savai Pullamsetti), as well as a number of other national and international research centers and clinics, to bring together the best possible expertise in the field of lncRNAs and PAH.
To identify hypoxia-relevant lncRNAs, pericytes and pulmonary artery smooth muscle cells from patients with idiopathic PAH were exposed to hypoxia and analyzed by RNA sequencing. Here, the lncRNA TYKRIL ("Tyrosine Kinase Receptor-Inducing lncRNA") was identified as the only lncRNA that consistently exhibited hypoxia-induced upregulation in all tissue and cell samples.
Knockdown of TYKRIL reversed the proproliferative and antiapoptotic phenotype induced under hypoxic and idiopathic PAH conditions.
Because TYKRIL was detected only in human cells because of the low species conservation of TYKRIL, ex vivo studies on human organotypic precision lung slices from patients with PAH were used for further investigations to approximate the in vivo situation. The use of precision lung sections from patients with PAH and the results obtained thereby represent an important milestone for translational research in PAH, as it greatly expands the possibilities for identification and characterization of novel and pathophysiologically relevant molecules expressed only in humans.
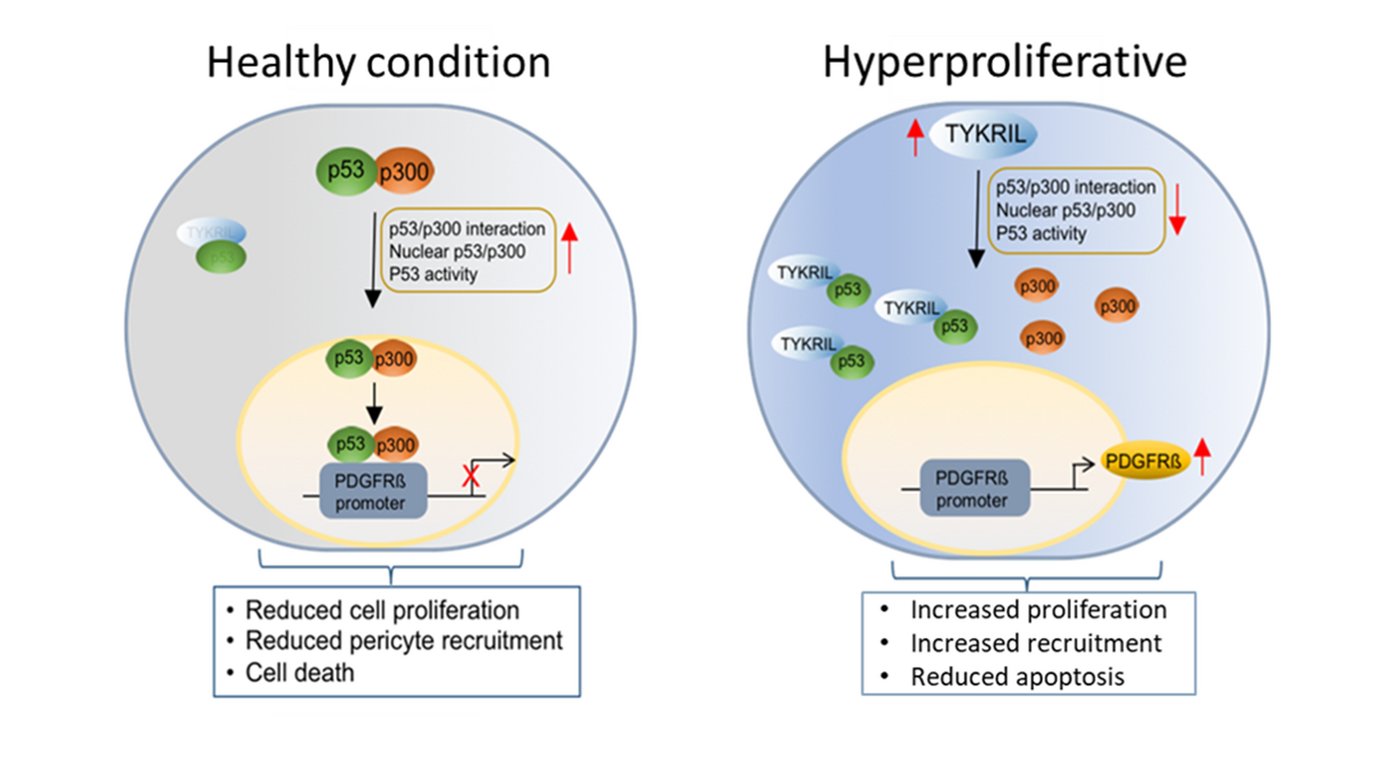
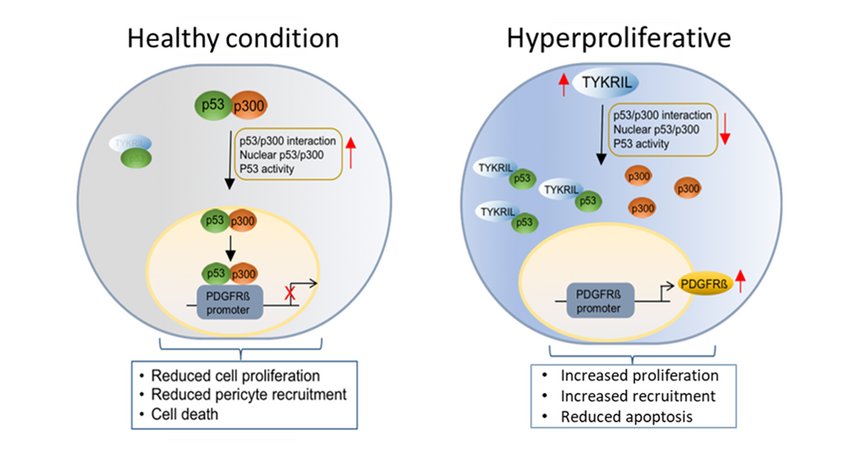
Inhibition of TYKRIL in precision lung sections reduced the vascular remodeling typical of PAH. The number of proliferating cells in the vessels was also reduced and the number of apoptotic cells in the vessels was increased compared with the control. Furthermore, a significant correlation of the expression of TYKRIL with platelet derived growth factor receptor beta (PDGFRβ), a key player in PAH, was found in PAH patient samples. Moreover, knockdown of TYKRIL significantly reduced PDGFRβ expression.
In further mechanistic studies, TKYIRL was shown to regulate the activity of the tumor suppressor p53, a known repressor of PDGFRβ. RNA immunoprecipitation experiments with various p53 mutants revealed that TYKRIL binds to the N-terminus of p53, which is an important binding region for the p53 co-activator p300. Interaction studies consequently revealed that TYKRIL blocks the p53-p300 interaction by binding to the N-terminus of p53. Thus, TYKRIL regulates p53 nuclear translocation in terms of a p53 "decoy" molecule.
As the first known lncRNA to regulate the p53/ PDGFRβ signaling axis, the previously uncharacterized lncRNA TYKRIL plays an important role in the pathogenesis of PAH and may represent a relevant target for future novel PAH therapeutic strategies.
Since 2004, the René Baumgart Foundation has been awarding an annual research award for scientific work that deals clinically or experimentally with the subject of pulmonary hypertension. The prize is endowed with 5,000 EURO.
You will find further information of the René Baumgart-Stiftung here (German only).