Signalling and mechanotransduction in the cardiovascular system
Project leaders: Dr. Paula Sofía Yunes Leites, Dr. Yongzhen Wei, Dr. Andy Cheng
PhD Students: Shenglan Zeng, Shangmin Liu, Yannick Jäger, Nimisha, Raphael, Yan Wang
Technical Assistants: Ulrike Krüger, Claudia Kopp

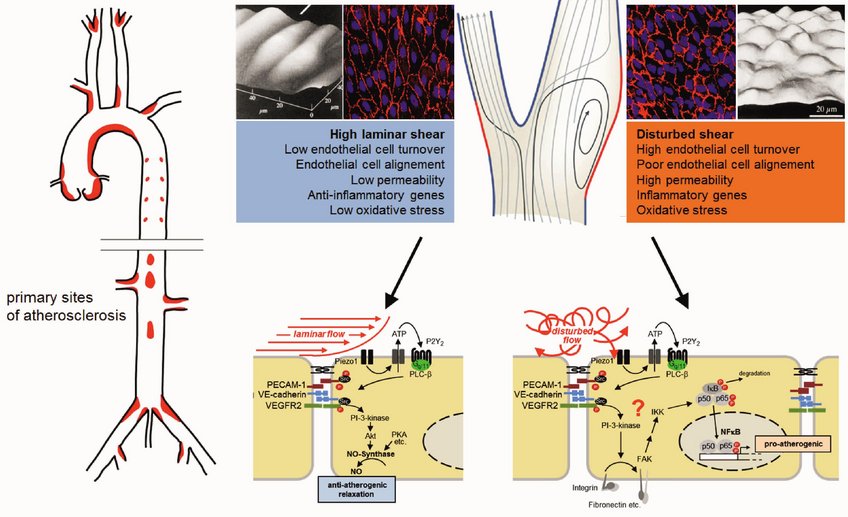
Fluid shear stress exerted by the flowing blood is crucial for the development of blood vessels but is also one of the major regulators of the vascular tone by inducing nitric oxide (NO) formation in endothelial cells, which relaxes vascular smooth muscle cells. Blood flow is also a key factor in the development of atherosclerosis, which mainly occurs in regions of arteries exposed to disturbances in fluid flow. Some of the endothelial mechanosensing and mechanotransduction mechanisms activated by laminar or disturbed flow to promote atheroprotective or atherogenic signaling have recently been identified. Understanding these fundamental processes provides new insights into the pathophysiology of cardiovascular diseases such as arterial hypertension and atherosclerosis.
Based on the analysis of basic signaling processes mediated by heterotrimeric G-proteins in vascular cells (Wirth et al., 2008; Korhonen et al., 2009, Althoff et al., 2012), we have identified some of the upstream mechanosensing and mechanosignaling mechanisms in endothelial cells, which involve the mechanosensitive cation channel Piezo1 and G-protein-coupled receptors (Wang et al., 2015; Wang et al., 2016; Albarrán-Juárez et al., 2018; Iring et al., 2019). Recent work aims at the identification of novel mechanosensitive and mechanotransducing processes in endothelial cells and other vascular cells which are differentially involved in anti-inflammatory (atheroprotective) and pro-inflammatory (atherogenic) signaling induced by laminar and disturbed flow, respectively. This includes the recently identified PKN2-mediated regulation of eNOS by laminar flow (Jin et al., 2021), the laminar flow-induced tenascin-X-mediated inhibition of endothelial-to-mesenchymal transition and atherosclerosis (Liang et al., 2022), the role of PIezo1 in integrating low flow and leukocyte adhesion to promote leukocyte extravasation (Wang et al., 2022) as well as the regulation of histone H3.3 phosphorylation by PKN1 to link disturbed flow-induced signalling to vascular inflammation and atherosclerosis. The ultimate goal is to better understand how endothelial dysfunction contributes to vascular diseases in order to identify new approaches to prevent cardiovascular disorders at an earlier stage as it is currently possible.
Literature
- Cho H, Lai CC, Bonnavion R, Alnouri MW, Wang S, Roquid KA, Kawase H, Campos D, Chen M, Weinstein LS, Martínez A, Looso M, Sanda M, Offermanns S. (2025) Endothelial insulin resistance induced by adrenomedullin mediates obesity-associated diabetes. Science. 387, 674-682.
- Jin YJ, Liang G, Li R, Wang S, Alnouri MW, Bentsen M, Kuenne C, Günther S, Yan Y, Li Y, Wettschureck N, Offermanns S. (2025) Phosphorylation of endothelial histone H3.3 serine 31 by PKN1 links flow-induced signaling to proatherogenic gene expression. Nat .Cardiovasc. Res. 4, 180-196.
- Li R, Shao J, Jin YJ, Kawase H, Ong YT, Troidl K, Quan Q, Wang L, Bonnavion R, Wietelmann A, Helmbacher F, Potente M, Graumann J, Wettschureck N, Offermanns S. (2023) Endothelial FAT1 inhibits angiogenesis by controlling YAP/TAZ protein degradation via E3 ligase MIB2. Nat Commun. 14, 1980.
- Wang S, Wang B, Shi Y, Möller T, Stegmeyer RI, Strilic B, Li T, Yuan Z, Wang C, Wettschureck N, Vestweber D, Offermanns S. (2022) Mechanosensation by endothelial PIEZO1 is required for leukocyte diapedesis. Blood 140: 171-183
- Liang G, Wang S, Shao J, Jin Y, Xu L, Yan Y, Günther S, Wang L, Offermanns S. (2022) Tenascin-X Mediates Flow-Induced Suppression of EndMT and Atherosclerosis. Circ Res. 130: 1647-1659
- Jin YJ, Chennupati R, Li R, Liang G, Wang S, Iring A, Graumann J, Wettschureck N, Offermanns S. (2021) Protein kinase N2 mediates flow-induced eNOS activation and vascular tone regulation. J Clin Invest. 131: e145734
- Iring A, Jin YJ, Albarrán-Juárez J, Siragusa M, Wang S, Dancs PT, Nakayama A, Tonack S, Chen M, Künne C, Sokol AM, Günther S, Martínez A, Fleming I, Wettschureck N, Graumann J, Weinstein LS, Offermanns S (2019) Shear stress-induced endothelial adrenomedullin signaling regulates vascular tone and blood pressure. J Clin Invest. 130: 2775-2791
- Chennupati R, Wirth A, Favre J, Li R, Bonnavion R, Jin YJ, Wietelmann A, Schweda F, Wettschureck N, Henrion D, Offermanns S. (2019) Myogenic vasoconstriction requires G12/G13 and LARG to maintain local and systemic vascular resistance. Elife 8
- Wettschureck N, Strilic B, Offermanns S (2019) Passing the Vascular Barrier: Endothelial Signaling Processes Controlling Extravasation. Physiol. Rev. 99: 1467-1525
- Albarrán-Juárez J, Iring A, Wang S, Joseph S, Grimm M, Strilic B, Wettschureck N, Althoff TF, Offermanns S. (2018) Piezo1 and Gq/G11 promote endothelial inflammation depending on flow pattern and integrin activation. J. Exp. Med. 215: 2655-2672
- Wang, SP, Chennupati R, Iring A, Kaur H, Wettschureck N, Offermanns S. (2016) Endothelial Piezo1 controls blood pressure by mediating flow-induced ATP release. J. Clin. Invest. 126: 4527-4536
- Wang S, Iring A, Strilic B, Albarrán Juárez J, Kaur H, Troidl K, Tonack S, Burbiel JC, Müller CE, Fleming I, Lundberg JO, Wettschureck N, Offermanns S (2015). P2Y2 and Gq/G11 control blood pressure by mediating endothelial mechanotransduction. J. Clin. Invest. 125: 3077-3086
- Althoff TF, Albarrán Juárez J, Troidl K, Tang C, Wang S, Wirth A, Takefuji M, Wettschureck N, Offermanns S (2012). Procontractile G-protein-mediated signaling pathways antagonistically regulate smooth muscle differentiation in vascular remodeling. J. Exp. Med. 209: 2277-2290
- Korhonen H, Fisslthaler B, Moers A, Wirth A, Habermehl D, Wieland T, Schütz G, Wettschureck N, Fleming I, Offermanns S (2009). Anaphylactic shock depends on endothelial Gq/G11. J Exp Med.
206: 411-420 - Wirth A, Benyó Z, Lukasova M, Leutgeb B, Wettschureck N, Gorbey S, Őrsy P, Horváth B, Maser-Gluth C, Greiner E, Lemmer B, Schütz G, Gutkind S, Offermanns S (2008). G12/G13-LARG-mediated signalling in vascular smooth muscle is required for salt-induced hypertension. Nat Med. 14: 64-68